 Recently, the single-atom alloys (SAAs) catalysts, consisting of exogenous isolated metal atoms dispersed on the surface of the metal host 23, 24, as a promising material with the advantages of high active atom utilization of single-atom catalysts (SACs) and alloy synergistic effect 25, 26, have been in the field of various electrocatalytic energy conversions 27, 28, 29, 30. To address these bottlenecks, it is fundamental to explore efficient bifunctional electrocatalysts for activating reaction pathways of the directed synthesis towards Ph-NH 2 and formate. Nevertheless, both Ph-NO 2-to-Ph-NH 2 and methanol-to-formate are multi-step reaction processes and are limited by numerous side reactions 21, 22. In view of this, the construction of Ph-NO 2 ERR-MOR coupled electrolytic system is a promising strategy for achieving high-value chemical synthesis from sustainable organic electrocatalytic conversion. Currently, the cheap and widespread methanol (CH 3OH, 350 USD/t) is considered an appropriate precursor for the high-value formate (HCOO −, 1300 USD/t) synthesis via anodic electrooxidation reaction 17, 18, and methanol electrooxidation reaction (MOR) coupled with various electrocatalytic reactions has attracted widespread research interest worldwide 19, 20. In contrast, the anodic biomass electrooxidation reaction not only can achieve the upgrading of cheap biomass but also offers the advantages of low energy consumption and high electrolysis efficiency 14, 15, 16. Notably, the anodic reaction for the conventional cathodic Ph-NO 2 electroreduction reaction (Ph-NO 2 ERR) is a sluggish kinetic oxygen evolution reaction (OER) 12, 13. Among the numerous organic electrocatalytic conversion reactions, the cathodic nitrobenzene (Ph-NO 2) electroreduction for aniline (Ph-NH 2) synthesis is regarded as a more low-carbon and environment-friendly green Ph-NH 2 synthesis process using H 2O and electrons as the hydrogen source and reductant, compared with high pollution and harsh conditions of traditional chemical synthesis method 8, 9, 10, 11. The organic electrocatalytic conversion, driven by electricity generated from renewable energy sources at ambient temperature and pressure 3, 4, is a green synthesis route for achieving controlled conversion of low-cost organic compounds into high-value chemicals and holds considerable research significance and application potential in the chemical industry and organic synthesis 5, 6, 7. The utilization of renewable energy is a crucial pathway for solving the increasing energy crisis and promoting the green low-carbon transformation of energy 1, 2. The synergistic catalytic effect and H*-spillover effect can improve catalytic reaction process and reduce energy barrier for reaction process, thus enhancing electrocatalytic reaction activity and target product selectivity. Density functional theory calculations reveal the electron effect between Cu single-atom and Rh host and catalytic reaction mechanism. In the coupled electrocatalytic system, the Cu single-atom-Rh metallene arrays on Cu foam requires only the low voltages of 1.18 V to reach current densities of 100 mA cm −2 for generating aniline and formate, with up to ~100% of nitrobenzene conversion/ aniline selectivity and over ~90% of formate Faraday efficiency, achieving synthesis of high-value chemicals. Here, we report a Cu single-atom dispersed Rh metallene arrays on Cu foam for cathodic nitrobenzene electroreduction reaction and anodic methanol oxidation reaction. In an oxidation half reaction, the electrons are products whereas in a reduction half reaction, the electrons are reactants.Organic electrocatalytic conversion is an essential pathway for the green conversion of low-cost organic compounds to high-value chemicals, which urgently demands the development of efficient electrocatalysts. The electrons are gained, thus, the half cell reaction at cathode is a reduction half reaction. 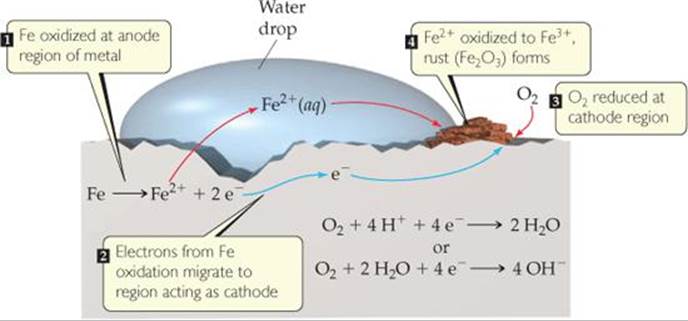
The example of a half cell reaction at anode is as follows: $$ ion gains 2 electrons and forms copper metal. The number of electrons lost and gained in the half cell reactions remains constant. Thus, the half cell reaction is one which takes place at one electrode. The two half cell reactions are collectively known as a redox reaction. 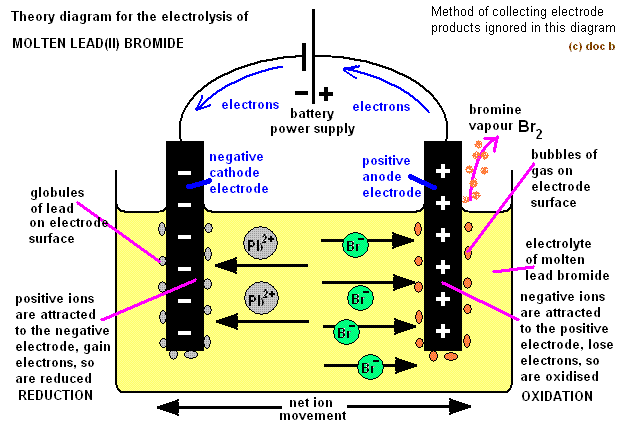
Ant at cathode, reduction reaction occurs, which is known as reduction half reaction. 
The half cell reaction occurs in an electrochemical cell at the electrodes.Īt anode, oxidation reaction occurs which is known as oxidation half reaction. In the oxidation reaction, electrons are lost and in the reduction reaction, electrons are gained. The half cell reaction occurs in an electrochemical cell.Ī half cell reaction can be an oxidation reaction or it can be a reduction reaction. Hint: An oxidation reaction in which the electrons are lost or a reduction reaction in which electrons are gained is known as a half cell reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
